AAPS PharmSci360 Short Course
Immunogenicity Risk Assessment for Biologic Proteins and Peptides
Saturday, November 3rd
1:00PM-5:00PM

Walter E. Washington Convention Center
801 Mt Vernon Pl NW
Washington, DC 20001
UPDATE:
Here are commemorative photos from our event. Thank you to all the speakers and participants for making our 2018 AAPS PharmSci360 Short Course a successful one!
- Annie De Groot, MD from EpiVax
- Sophie Tourdot, PhD from Pfizer
- Shibani Mitra-Kaushik, PhD from Sanofi
- Brian Roberts, PhD from EpiVax
- Vibha Jawa, PhD from Merck
- Amy Rosenberg, PhD from FDA
Join EpiVax a day early at AAPS PharmSci360 on Saturday, November 3rd from 1:00PM-5:00PM, as we host our very own pre-conference short course: “Immunogenicity Risk Assessment Update for Biologic Proteins and Peptides.”
Protein therapeutics hold a prominent and rapidly expanding place among medicinal products, however unwanted immunogenicity revealed at a late stage can derail long and costly development programs. Despite early belief that the fully human nature of some therapeutic proteins such as recombinant cytokines, growth factors, enzyme replacement factors, and monoclonal antibodies would represent a significant advantage, adverse effects associated with immune responses to some biologic therapies have become a topic of some concern. As a result, drug developers are devising risk mitigation strategies to assess immune responses to protein therapeutics during both the pre-clinical and the clinical phases of development. While there are many factors that contribute to protein immunogenicity, T cell- (thymus-) dependent (Td) responses appear to play a critical role in the development of antibody responses to biologic therapeutics.
Our speakers will provide a timely update, describing updates to a range of methodologies to predict and measure Td immune responses to protein drugs. The benefits and limitations of current in-silico and in-vitro assay methods, in vivo animal models, including different humanized mouse formats in the field of Immunogenicity Risk Assessment, will also be touched upon.
This session will feature the following discussions:
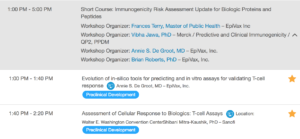

To learn about all the pre-conference activities taking place on Saturday, November 3rd and Sunday, November 4th, 2018, please visit the AAPS PharmSci360 official event page.
CHECK BACK FOR PROGRAM UPDATES!