Press
New Analysis of 99 Ebola Sequences Published August 29th
Posted on August 30, 2014 by Annie De GrootMatt Ardito (EpiVax IMF team-member) did a quick analysis of the new Ebola sequences on Friday August 29th. He downloaded the newly published Ebolavirus GP proteins from GenBank. After loading the new sequences (from https://www.ncbi.nlm.nih.gov/bioproject/PRJNA257197) into IVAX, he selected the 16 original Ebola clusters that EpiVax immunoinformatician Guilhem Richard (IMF team-member) found in his ...Read moreEbola – Extending our Hand to Help
Posted on August 17, 2014 by Annie De GrootEbola - Extending our Hand to HelpRead moreNew Immunogenicity Analysis Tool: CHOPPI
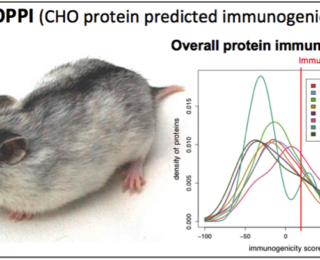
Posted on July 8, 2014 by Annie De GrootNew Immunogenicity Analysis Tool Emerges from Dartmouth-EpiVax-URI Collaboration “In Silico” Host Cell Protein for Protein Therapeutics Providence, RI. Tuesday, July 8th, 2014 A Public-private partnership resulted in the development of a new web-based tool that will help manufacturers of protein-based therapeutics improve the safety of their manufacturing processes, avoiding problems that caused the FDA to suspend a ...Read moreCV4 Submission Deadline Extended!
Posted on June 24, 2014 by Annie De GrootWe are pleased to announce an extension of the deadline to submit full-length papers to the 4th Computational Vaccinology Workshop. The deadline, originally July 1st will be extended to August 4th. The deadline for poster abstracts is August 25th. Hopefully this extra time will allow you to polish your submissions to this exciting workshop. For more details ...Read moreNEWS: Vaccines are not associated with autism
Posted on May 21, 2014 by Annie De GrootVaccines are not associated with autism: An evidence-based meta-analysis of case-control and cohort studies Luke E. Taylor, Amy L. Swerdfeger, Guy D. Eslick (Not an EpiVax publication) Highlights • Five cohort studies involving 1,256,407 children and five case-control studies involving 9920 children were included in this analysis. • There was no relationship between vaccination and autism (OR: 0.99; 95% CI: 0.92 to ...Read moreEpiVax Immunogenicity Seminar – September 17th 2014 at the Westin Tokyo
Posted on May 12, 2014 by Annie De GrootEpiVax will be in Tokyo Japan from September 14th-20th, 2014 hosting our semiannual immunogenicity seminar and meeting with collaborators. The Westin Immunogenicity Seminar is scheduled for September 17th, 2014. The purpose of this seminar is to bridge the gap between recent academic research on Tregs, Tregitopes and protein drug development (immunogenicity, deimmunization and tolerance induction). Links PDF Agenda Last year’s event ...Read moreDe Groot to speak at ASCLS-CNE event
Posted on April 24, 2014 by Annie De GrootAPRIL 24, 2014 EpiVax CEO/CSO, Dr. Annie De Groot MD, will be one of the featured speakers during next week’s 66th Annual Clinical Laboratory Science Convention. The event is scheduled for April 29 through May 1 at the Rhode Island Convention Center in Providence. Dr. De Groot will speak on Tuesday, April 29 during a workshop on ...Read moreURI & EpiVax scientists – closer to designing vaccine for H. pylori pathogen
Posted on April 21, 2014 by Annie De GrootProvidence, R.I. – April 18, 2014 – Researchers from the University of Rhode Island and EpiVax, Inc. are championing a recent breakthrough in the laboratory with the hopes it could lead to a vaccine against the pathogen responsible for stomach cancer and other ailments. The results are published today in the Public Library of Science One ...Read moreImmunogenicity and Tolerance Seminar – Amsterdam 2016
Posted on April 1, 2014 by Annie De GrootPlatine and EpiVax are co-organizing their 2nd seminar on Immunogenicity and Tolerance on October 24th, 2016. Click this link to register This meeting will focus on the determinants of immunogenicity – a key feature of effective vaccines and a key barrier to success in the clinic for protein therapeutics. Conversely, tolerance induction is a key feature of successful protein therapeutics, while ...Read moreICoVax 2013 papers are now published
Posted on March 19, 2014 by Annie De GrootIcoVax Update: The publications from our “ICoVax 2013” pre-conference have now been published. The full content of the supplement can be found at this link. The link to the supplement is permanently accessible. We hope to see you at the next event! ICoVax description: The annual Vaccine & ISV Annual Global Congress, co-organized by the Vaccine journal and the International ...Read moreNow in Malaysia: "Stealth" virus H7N9
Posted on February 13, 2014 by Annie De GrootFrom CIDRAP: Stealth Virus Moves in on Malaysia. This is the first case reported outside of China/Hong Kong; more are expected. The case fatality rate is currently at 24%. Health officials announced an H7N9 avian flu infection in Malaysia today, the first case detected outside of China, along with eight other newly confirmed cases—one in Hong ...Read moreScience: Avian Flu Concerns Shutter Poultry Markets
Posted on January 29, 2014 by Annie De GrootAvian Flu Concerns Shutter Poultry Markets 28 January 2014 4:15 pm 0 Comments M M (Padmanaba01)/Wikimedia CommonsFlu source? A poultry market in China. WASHINGTON, D.C.—With human cases of H7N9 avian influenza piling up, Chinese authorities have ordered the closure of live poultry markets in three eastern cities in a bid to stem transmission of the virus. So far, the Chinese ...Read morePBN: New research, same mission: ‘improve human health’
Posted on January 22, 2014 by Annie De GrootNew research, same mission: ‘improve human health’ EPI CENTER: EpiVax founder and CEO Anne S. De Groot described herself as a “physician to make products that help human health everywhere.” PBN PHOTO/MICHAEL PERSSON By Harold Ambler Contributing Writer 1/13/14 If “immunoinformatics” ever becomes a household word, the team at Providence’s EpiVax Inc., led by CEO Dr. Anne S. De ...Read more. . . #ToldYouSo – EpiVax H7N9 ‘Stealth Virus’ Prediction a Slam Dunk
Posted on January 4, 2014 by Annie De GrootAdd two previous reports of the low immunogenicity of H7N9 HA, one from NovaVax, stating that their unadjuvanted vaccine is only 6-16% effective and Novartis – showing only 6% seroconversion for their unadjuvanted HA vaccine) to this report – and you have a slam dunk – three independent verifications that our tools accurately predict vaccine ...Read moreEmployment Opportunities at a Cutting Edge Biotech Company in Providence
Posted on January 4, 2014 by Annie De GrootWhy work at EpiVax? Members of the EpiVax team strive to see beyond the horizon, desiring to be part of an effort that is bigger than themselves by contributing to improving human health by moving science forward. In the long term, we aim to leave behind a legacy for the next generation, and we therefore continually ...Read more
Join our newsletter
We strive to provide our clients, collaborators and everyday immunology enthusiasts with valuable content. Written by our CEO/CSO Dr. Anne Dr Groot, you can expect up to date vaccine development efforts, upcoming events and so much more from our offices here in Providence, RI.