EpiVax Inc. enters license agreement with Novozymes Biopharma to advance pioneering treatment of autoimmune diseases
Novozymes’ Veltis® albumin-based half-life extension platform will help move Tregitope treatment for autoimmune diseases closer to clinical development.
Providence, RI. Monday December 22, 2014 EpiVax and Novozymes Biopharma today announced that they have entered a license agreement for the development of novel treatments for autoimmune diseases. The agreement follows two years of successful feasibility studies and it allows EpiVax to use Novozymes’ albumin technologies to develop cutting edge immunotherapies.
EpiVax has identified a set of natural peptide sequences, called Tregitopes, derived from Immunoglobulin G (IgG) that are potent activators of natural T regulatory cells (hence Tregitope). The initial application targeted by the Novozymes/EpiVax collaboration will be the development of a recombinant alternative to intravenous IgG (IVIG) treatment, a global market that exceeded $6 billion in 2014.
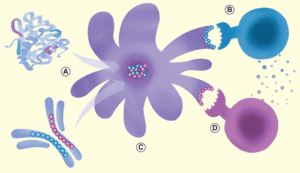
Tregitopes are naturally occurring T cell epitopes found in IgG. They can be inserted into proteins or delivered in albumin fusions (a). Once processed by antigen presenting cells, they induce Tregs to be activated (b). These activate nTregs induce tolerogenic antigen presenting cells (c) and suppress nearby T effector inflammatory responses (d).
Tregitopes will be fused to albumin, a naturally occurring protein that can be found in human blood. Albumin works as a vehicle that transports the therapy throughout the body and increases the lifespan of the therapy. The albumin is produced by Novozymes and known as VELTIS®.
“We are delighted to move into a license agreement with EpiVax in this key area of autoimmune diseases treatment”, says Peter Rosholm, Vice President at Novozymes Biopharma. “In combination with the technologies and know-how EpiVax holds, Novozymes’ VELTIS makes it possible to develop a potential ‘paradigm-shifting’ treatment for autoimmune diseases.
Treatment of autoimmune diseases
Tregitopes were discovered in 2008 by the team of De Groot and Martin at EpiVax; the original discovery was published in the journal Blood in 2008[1]. Tregitopes act as a natural immune system modulator and have been shown in standard preclinical models, and by collaborating laboratories, to suppress and treat autoimmune disease, allergy, and to effectively suppress the immunogenicity of co-administered proteins. Research at EpiVax has generated substantial evidence that Tregitopes may explain one of the mechanisms by which IVIG acts to treat autoimmune disease.
“Novozymes’ proven solutions, technical expertise, and reputation as a reliable partner, makes it an ideal company for us to work with as we move our Tregitope immune-modulating technology even closer to clinical trials. After successful feasibility studies, we now move into the next phase, looking to develop a platform for immunomodulation that will change the way that we treat autoimmune diseases, allergy and transplantation, says Dr. Annie De Groot, CEO/CSO at EpiVax, Inc.
Novozymes has also recently announced a number of collaborations with other pharmaceutical companies. These companies can now evaluate Novozymes’ albumin technology for potential new drug candidates. Earlier this year, GlaxoSmithKline achieved market approval for their new diabetes drug based on Novozymes’ albumin technology.
“We consider this new license agreement yet another proof that VELTIS can offer true benefits for our customers seeking to develop longer lasting and better drugs”, says Peter Rosholm.
[1] De Groot A.S., et al., Activation of Natural Regulatory T cells by IgG Fc-derived Peptide “Tregitopes”. Blood, 2008,112: 3303. https://tinyurl.com/ASDeGroot-Blood-2008
