March of the Teenage B Cells
BCR are Like Teenagers

I remember teenagers. I suppose that the last phase of teen development prepares you for their departure, and then they are gone… Hopefully, the memories of their sweet, former personalities will last longer than the drama.
Similarly, antibodies (a.k.a BCR when attached to B cells) move from sluggish to reactive (immunologically) as they mature (through somatic hypermutation (SHM)). It turns out that SHM isn’t just about fine-tuning binding affinity, it’s also about what gets left behind. Our new Frontiers paper takes a closer look at how SHM might increase immunoreactivity, specifically by reducing Treg epitopes (Tregitopes™) while sparing potential effector T cell epitopes. I think that’s amazing.
What does that mean for therapeutic antibody design? Let me know what you think! It’s in proofs as I write this, but you can sign up to get the paper at the link.
Smoothing the ANDA Pathway
. . . It’s Orthogonal!
The latest Epi-paper on Generic Peptides, published in Pharmaceutical Research, dives into how the PANDA® approach at EpiVax helps assess the immunogenicity of peptide drugs and their impurities, a key step in obtaining FDA approval through the ANDA pathway.
The paper reviews in silico and in vitro methods and compares these to other methods like MAPPS assays. We provide several case studies, recent insights and best practices for tackling immunogenicity concerns in generic peptide drugs. The goal of this publication and the PANDA® Screening program is to make regulatory filings much easier for generic peptide drug developers.
. . . CTO Masters the Master File
Our new CTO Guilhem Richard has officially submitted a Model Master File (MMF) for the PANDA® process to the FDA. This resource ensures that the science behind PANDA is accessible to FDA reviewers. Generic drug developers can refer to the MMF (contact us if you need a letter), and the FDA gets direct access to the data that supports the immunogenicity models. It’s a Win-Win!
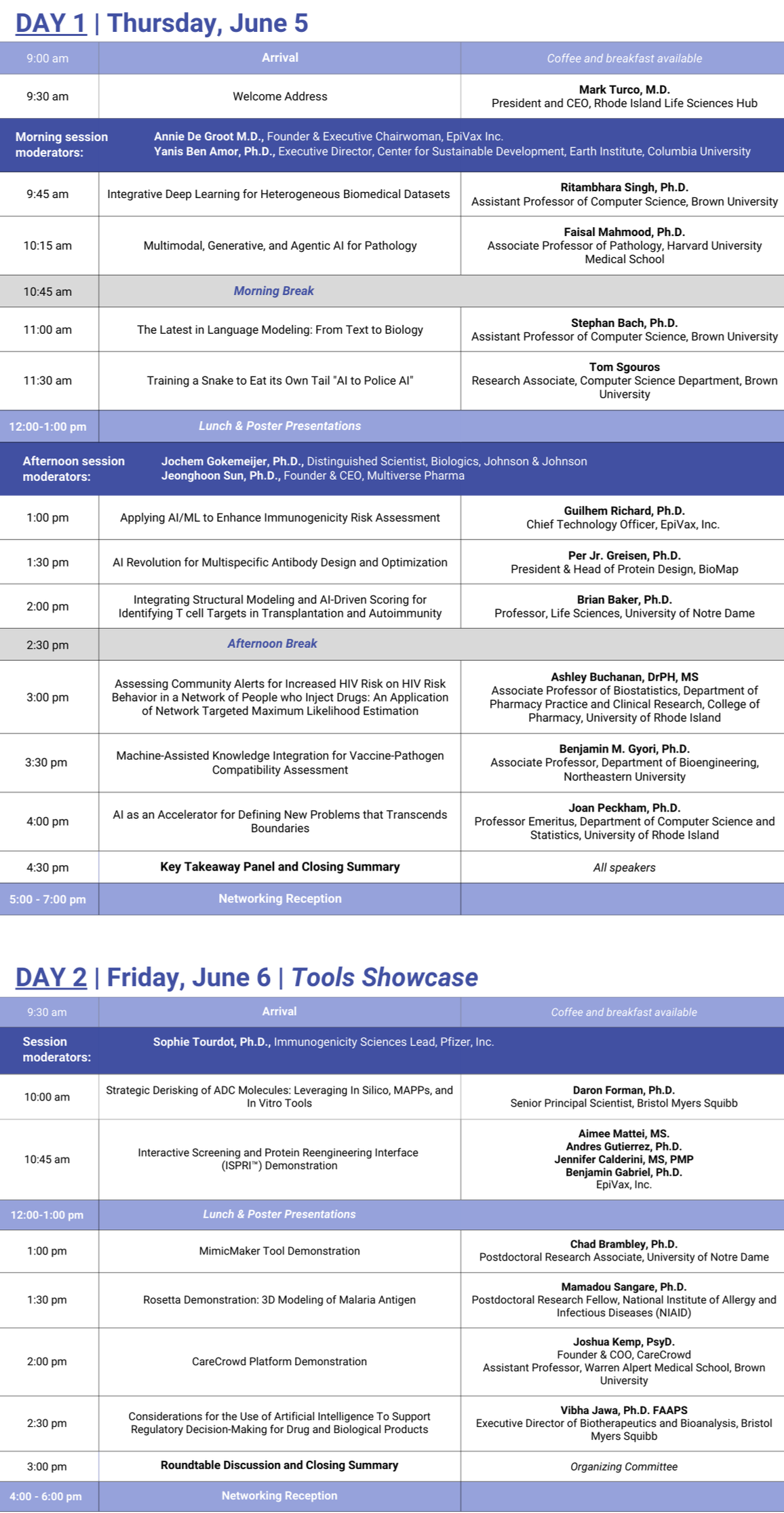
AI / HI is Back!

Registration is open now (and free!) — sign up to reserve your spot and to get important updates. More information here.