Confirm and contextualize in silico predictions with human-relevant in vitro insights.
Inform mitigation, comparability, and formulation decisions with experimental data.
Support regulatory-aligned immunogenicity risk assessment strategies
De-Risk Development with In Vitro Immunogenicity Insights.
Integrating in vitro immunogenicity assessments into the drug product development strategy is essential for thorough validation. While in silico analyses offer initial insights into candidate sequence immunogenicity, in vitro assays are orthogonal methods that can be used to not only confirm findings but also address additional product-related immunogenicity concerns.
EpiVax provides comprehensive in vitro immunogenicity screening services to guide therapeutic and vaccine assessment and optimization. This data provides a strong foundation for later-stage decision making, helping developers anticipate immunogenicity risks and inform bioanalytical and clinical strategies.

Cytokine Storm Assay
Innate Assay
PBMC Assay
DC:PBMC Assay
HLA Binding Assay
Assays for Assessment of Generic Peptides
Cytokine Storm Assay
Evaluates potential stimulation of excessive cytokine release from human whole blood samples following exposure to a test article using multiplexed cytokine analysis (Luminex). The assay provides insights regarding a product’s ability to induce immune overstimulation and offers a mechanistic view of early immune activation.
What to assess: Biologics (antibodies, ADCs, cytokine-based therapies, etc.), peptides, proteins, and cell and gene therapies with a mechanism of action that have a direct or indirect immunomodulatory function.
Input: Whole blood
Incubation: 1 day
Innate Assay
Measures cytokine release resulting from engagement of innate immune receptors in human whole blood or peripheral blood mononuclear cell (PBMC) samples following exposure to a test article, using multiplexed cytokine analysis (Luminex).
What to asses: Immune cell engagers, impurities, viral vectors, nucleic acids, product formulations.
Input: Whole blood, PBMC
Incubation: 1 day
PBMC Assay
Measures innate and/or adaptive immune responses of human PBMCs following exposure to a test article using multiplexed cytokine analysis (Luminex), cell proliferation (flow cytometry), and/or ELISpot. As a modular “base” assay, the design can be customized for scale, donor number, readouts, and benchmark controls to match study goals (i.e. to support earlier stage lead selection/ optimization/ characterization or later stage product impurity evaluation, product lot comparisons, and clinical response justifications for regulatory agencies).
What to assess: Biologics, peptides, impurities, products, etc. that are not expected to have an immunomodulatory mechanism of action.
Inputs: PBMC
Incubation: 1-7 days
DC:PBMC Assay
Measures the adaptive immune responses of human PBMCs following co-culture with test article- ‘loaded’ dendritic cells (DCs) using multiplexed cytokine analysis (Luminex), cell proliferation, and/or ELISpot. The assay is key to evaluating the immunogenicity risk of a molecule with a defined immunomodulatory mechanism but otherwise has similar uses to the PBMC assay described above. Useful in later stages for immunogenicity risk re-assessment, including evaluation manufacturing changes and confirming consistency of immunogenicity profiles.
What to assess: Biologics, peptides, products, etc., with known immunomodulatory mechanism of action.
Inputs: DC/PBMC co-culture
Incubation: 7-10
HLA Binding Assay
Measures the relative binding affinity of peptides or peptide regions derived from protein sequences to a panel of recombinant Class II HLA alleles, using a fluorescence-based readout that is inversely proportional to the test article’s binding affinity. This cell-free assay is commonly used to validate in silico HLA binding predictions by confirming peptide affinity for HLA, a prerequisite for T cell activation, as the assay includes a panel of Class II HLA that mirror those modeled in EpiVax in silico assessments. It is also a valuable first-line approach for peptides containing high levels of unnatural amino acids that may not be suitable for computational analysis.
What to assess: Peptides or peptide regions of proteins, complex biologics, etc.
Input: Recombinant HLA alleles
Incubation: 2 days
PANDA® Screening
Designed for sponsors pursuing the Abbreviated New Drug Application (ANDA) Pathway for generic peptide drugs.
The FDA recently released guidance requiring sponsors pursuing the Abbreviated New Drug Application (ANDA) pathway to identify and characterize process and product-related impurities in their drug formulations.
In response, EpiVax adapted its proprietary in silico and in vitro methods for assessing immunogenic risk to offer Peptide Abbreviated New Drug Application (PANDA®) Screening – a set of orthogonal analyses tailored to specifically meet ANDA sponsors’ adaptive and innate immunogenicity assessment needs for generic peptides and their related impurities.
In vitro assays included in the PANDA® Screening program include:
Class II HLA Binding Assay:
Measures the relative binding affinity of API and impurity peptides (or putative T cell epitope regions of the sequences) to Class II HLA alleles
T cell Assay:
Tests the ability of API and impurity peptides to stimulate a T cell response using human peripheral blood mononuclear cells (PBMC).
Innate Immune Response Assay:
Used to assess innate immunogenicity of products using human PBMCs.
ISPRI Design™
To prepare for in vitro studies, the EpiVax experts can design peptides for synthesis using our advanced in silico tools.
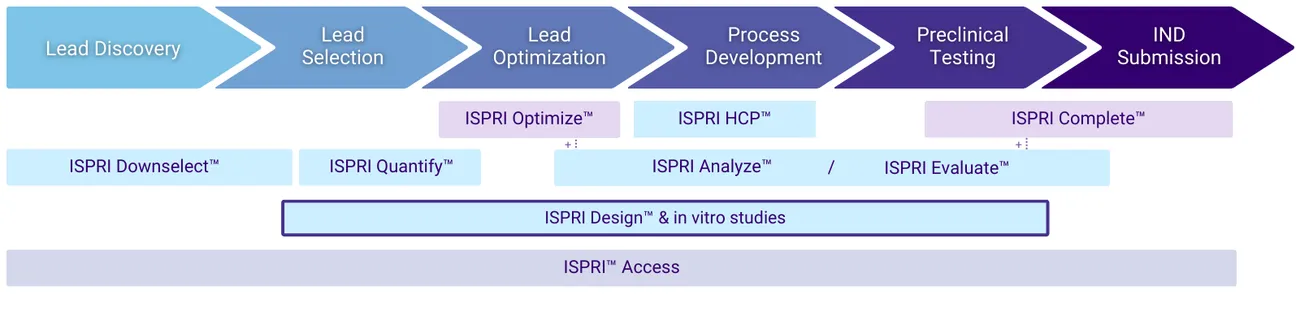
Where are in vitro assessments applied during the development lifecycle?
Lead Optimization
Deimmunization of High-Risk Sequence Regions
Test peptide regions with suggested modifications against relevant controls and a small number of blood donors to rank.
Lead Characterization
Detailed Assessments for IND Preparation & More Comprehensive Risk Profiling
Test whole molecules against relevant controls and a large number of blood donors to inform regulatory submissions and clinical strategy.
Risk Re-Assessment
De-risking of Manufacturing Changes
Confirm the consistency of your therapeutic’s immunogenicity risk profile when scaling from clinical lots to commercial scale manufacturing.
Need human-relevant immunogenicity data to support development decisions? Get in touch!
The EpiVax Roadmap