
Smarter vaccine design will circumvent immunoregulatory evasion in chronic HIV and HCV infection
In: Why vaccines to HIV, HCV and Malaria have so far failed – challenges to developing vaccines against immunoregulating pathogens. Frontiers in Microbiology. 2014. Editor (Gowans).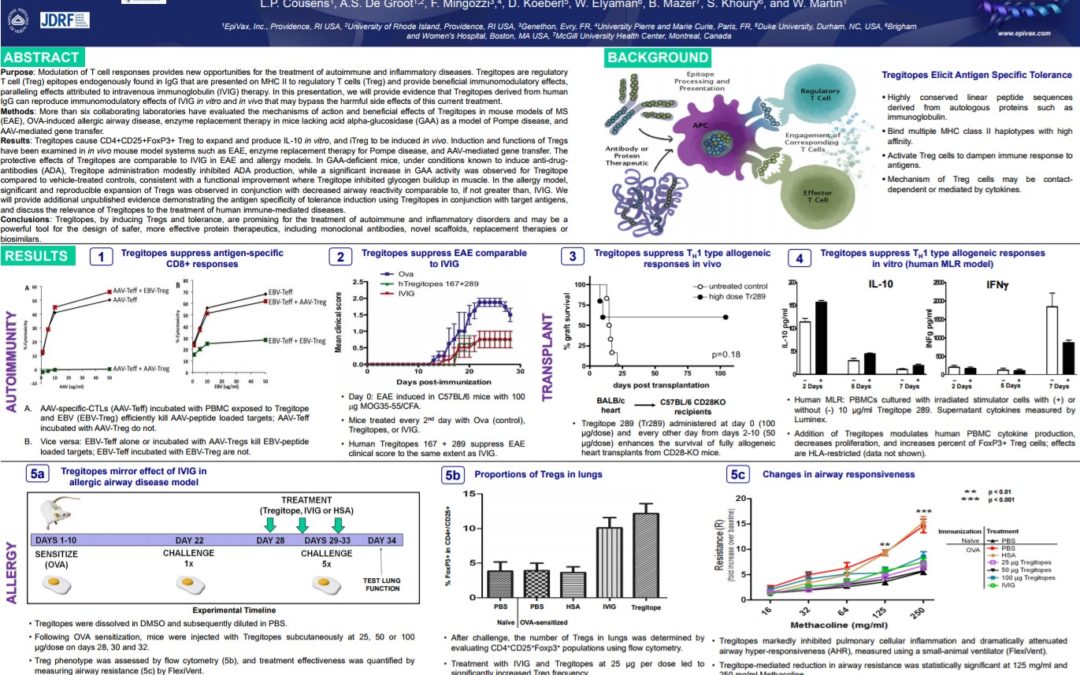
Tregitope Applications to Tolerance Induction in Autoimmune Disease and Therapeutic Protein Immunogenicity
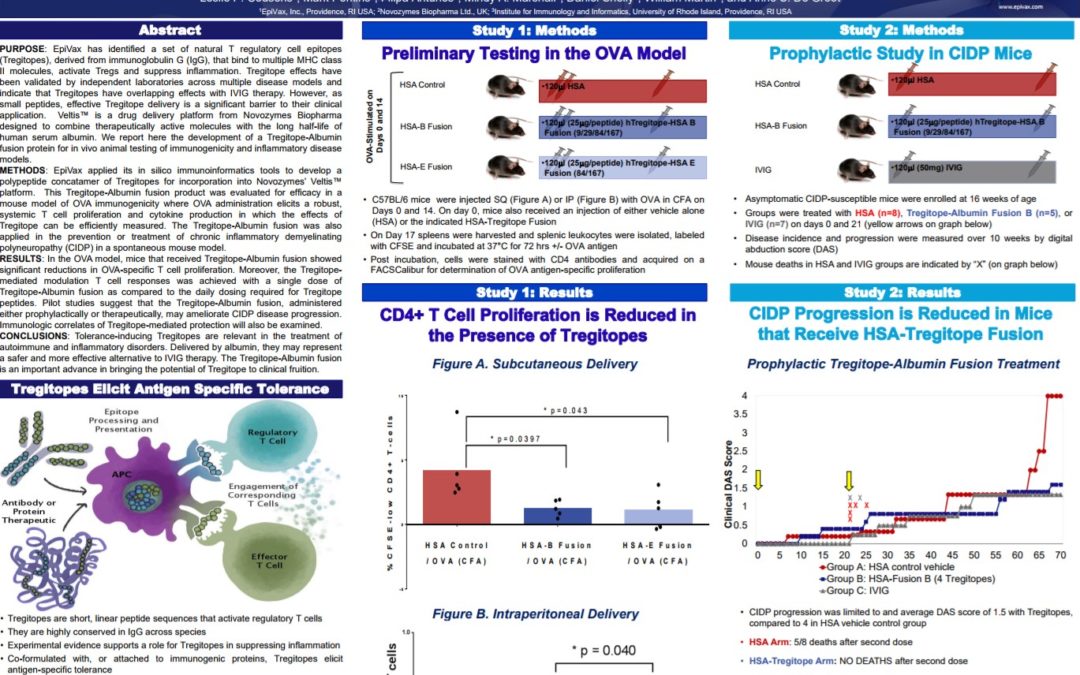
Albumin Delivery of Tregitope Peptides for Tolerance Induction in Autoimmunity and Inflammatory Disease
