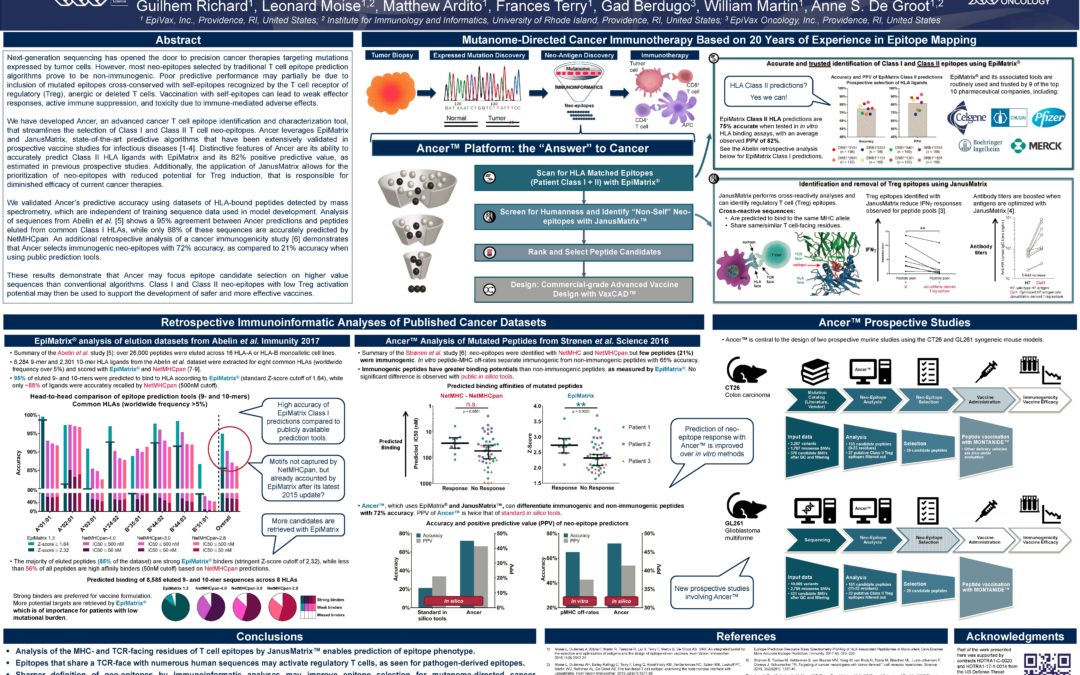


New Regression Model Predicts Antibody Immunogenicity Based on Effector and Regulatory T Cell Epitope Content
EpiVax_AET18_New Regression Model Predicts Antibody Immunogenicity_5Dec18
From infectious disease to personalized cancer vaccines: Regulatory T cell epitopes determine patient outcomes and suppress neoantigen responses
EpiVax_Oncology_Ancer_SITC_05Nov18_39Wx35H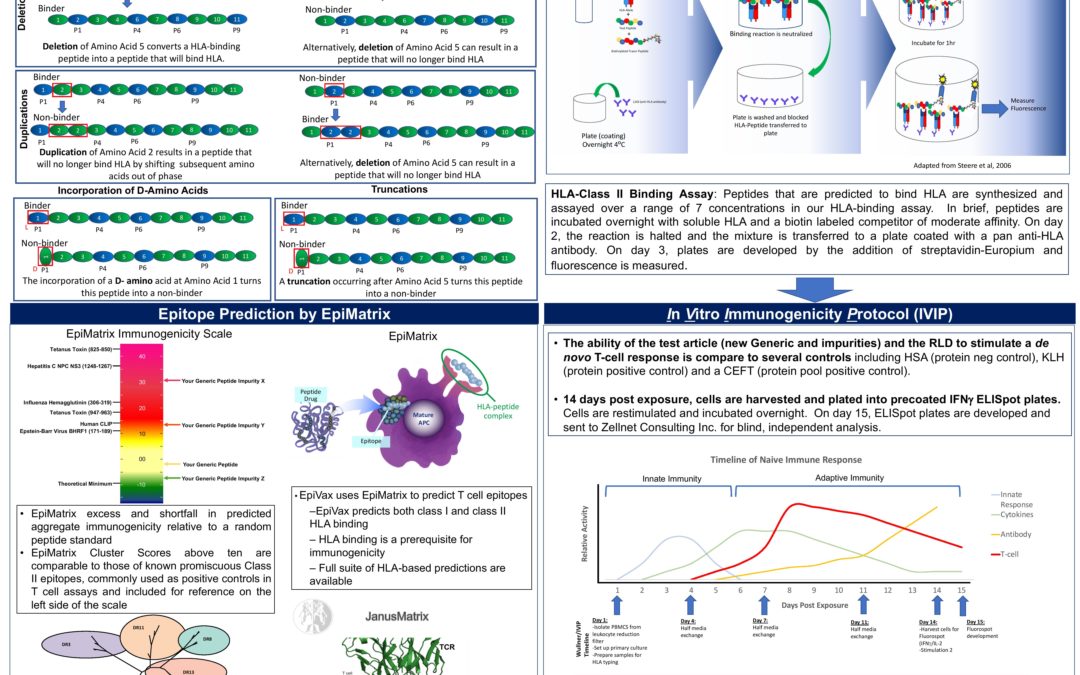
Innovative Preclinical Assessment Tools for Safety and Efficacy of Protein and Peptide Therapeutics…of Peptides and P-ANDAS
EpiVax_EuroTIDES_PANDA_02Nov18 To learn more about EpiVax’s PANDA Program, read our latest entries. PANDA screening Interview with Brian Roberts: Protein Therapeutics Manager PANDA Press Release