
Development of highly stable and de-immunized versions of recombinant alpha interferon: Promising candidates for the treatment of chronic and emerging viral diseases
Development of highly stable and de-immunized versions of recombinant alpha interferon: Promising candidates for the treatment of chronic and emerging viral diseases
T-cell dependent immunogenicity of protein therapeutics: Preclinical assessment and mitigation
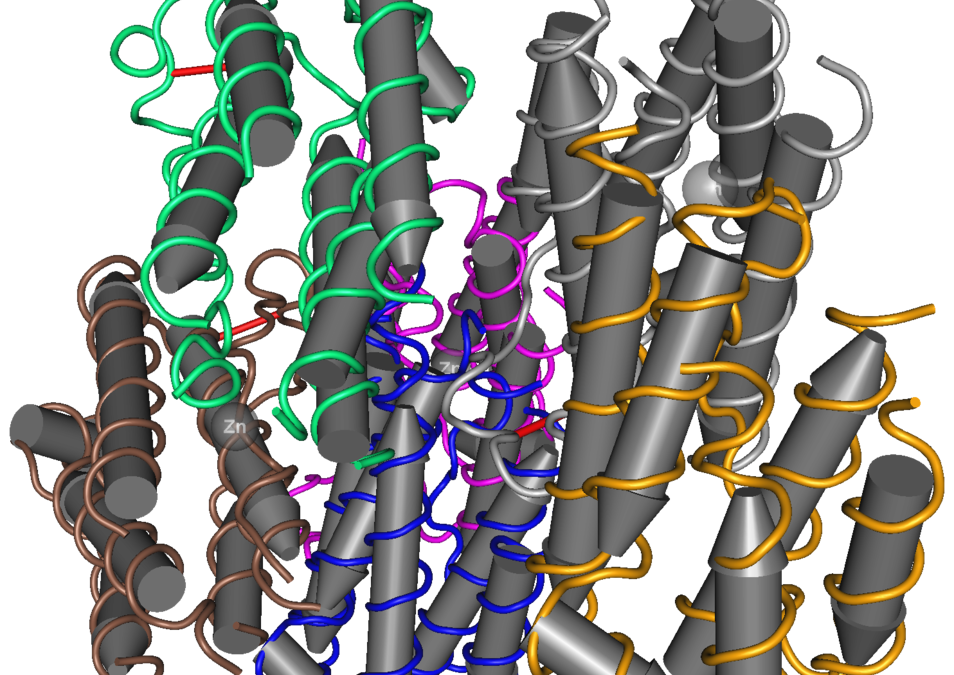
De-immunized and Functional Therapeutic (DeFT) versions of a long lasting recombinant alpha interferon for antiviral therapy
