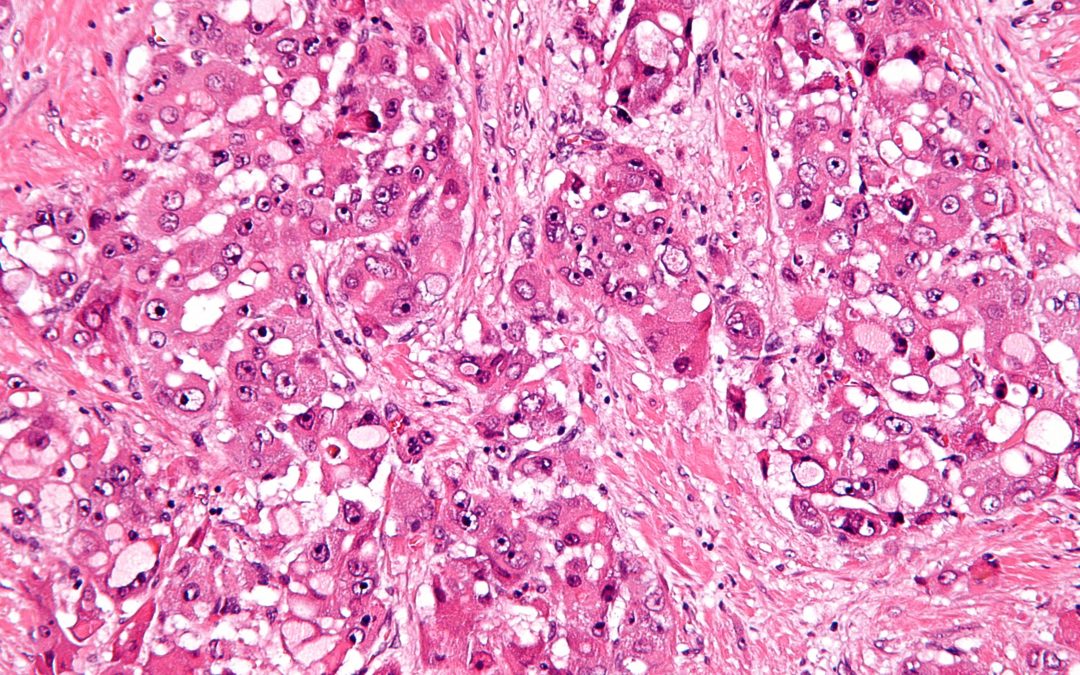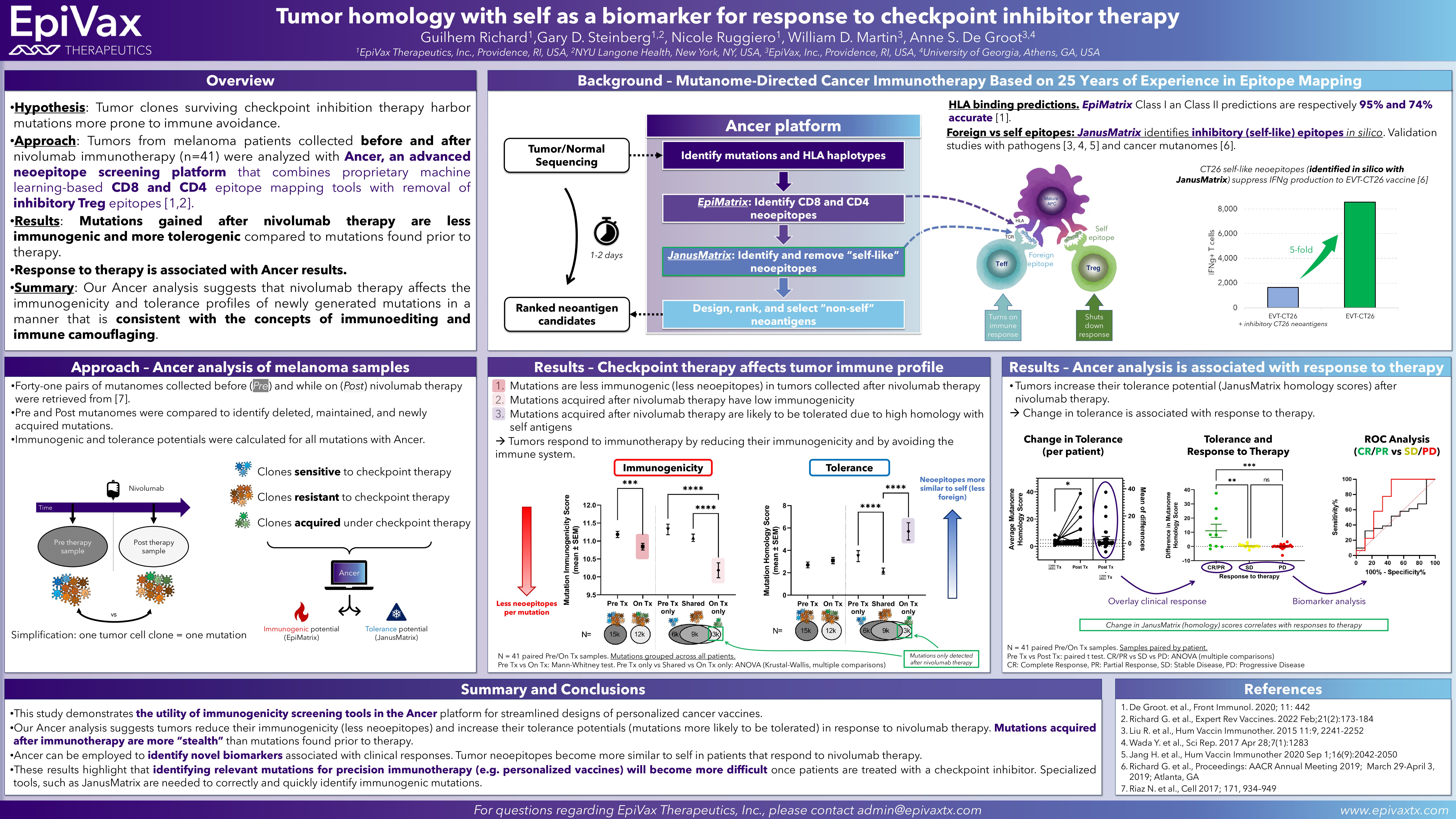

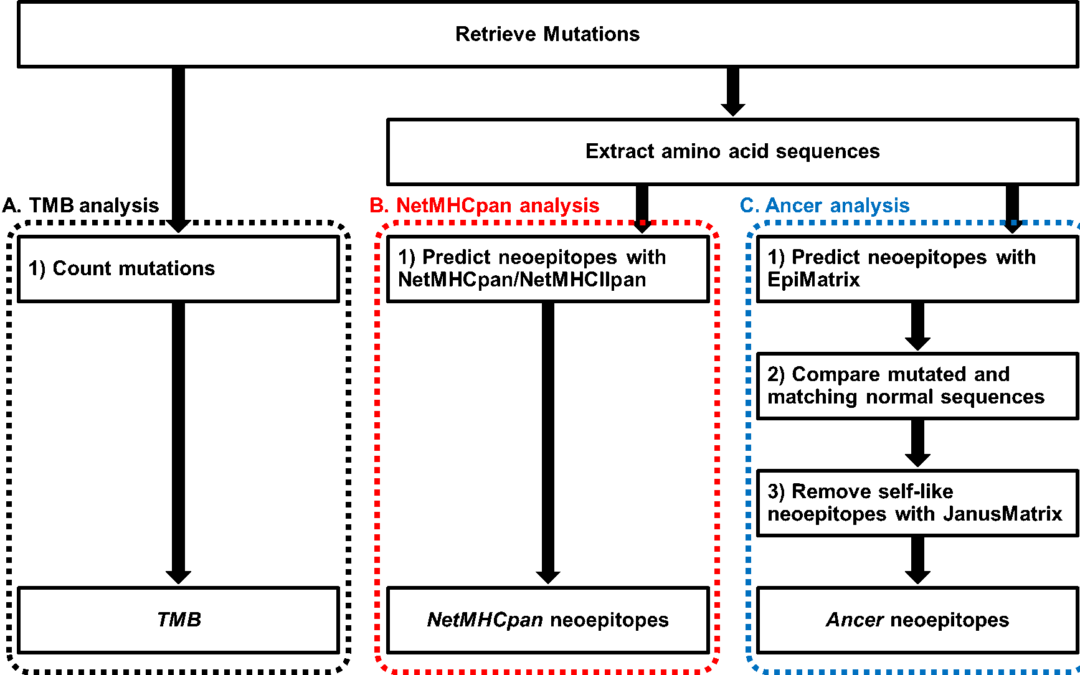
Multi-step screening of neoantigens’ HLA- and TCR-interfaces improves prediction of survival
A fibronectin scaffold approach to bispecific inhibitors of epidermal growth factor receptor and insulin-like growth factor-I receptor
A fibronectin scaffold approach to bispecific inhibitors of epidermal growth factor receptor and insulin-like growth factor-I receptor. Adnexus MAbs. 2011 [ISPRI prediction of immunogenicity]
Bioinformatics tools for identifying class I-restricted epitopes
Methods (Epitope Mapping Issue). Bill Kwok, editor, Methods 29 (2003) 289–298.