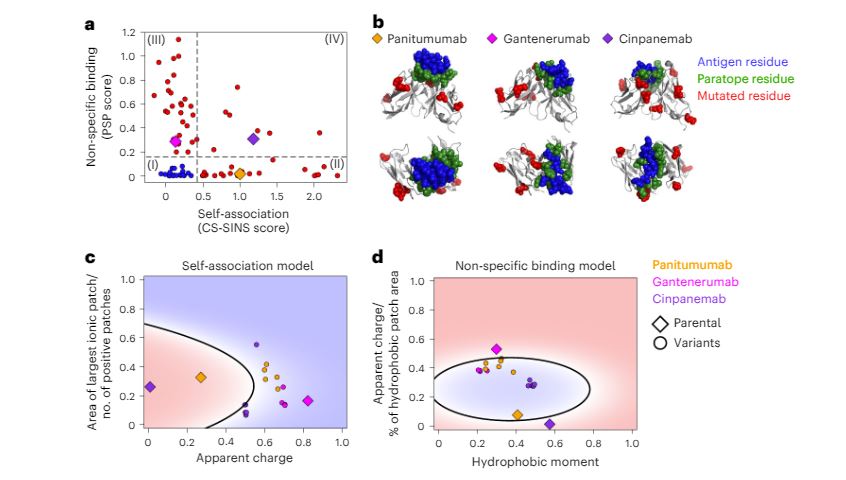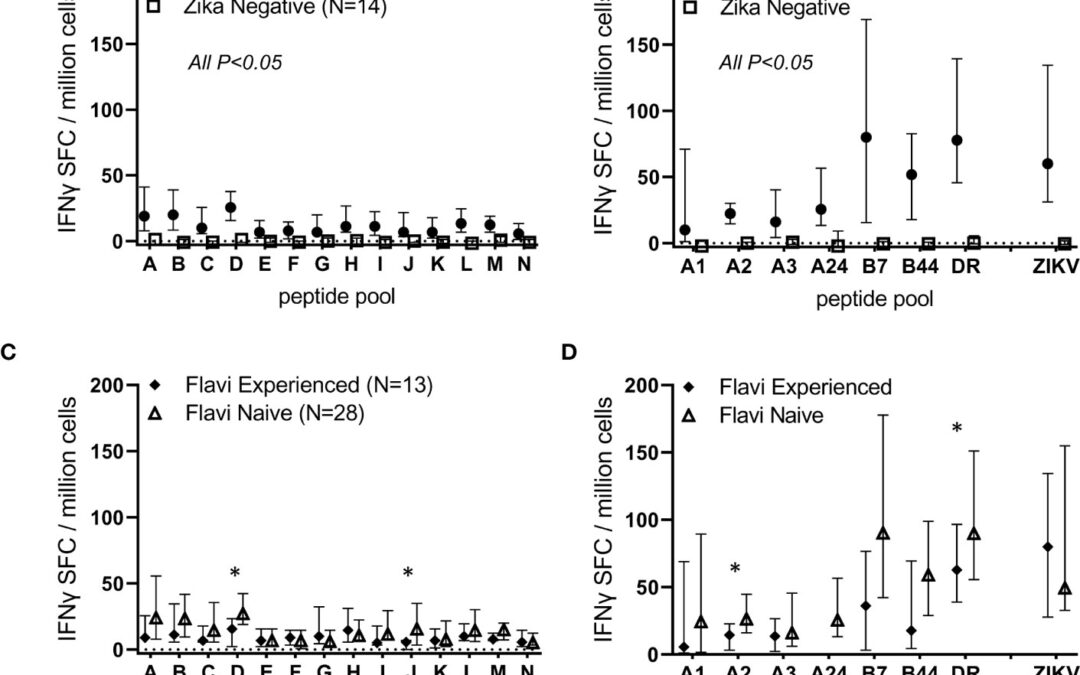
by Elena Iemma | Sep 21, 2023
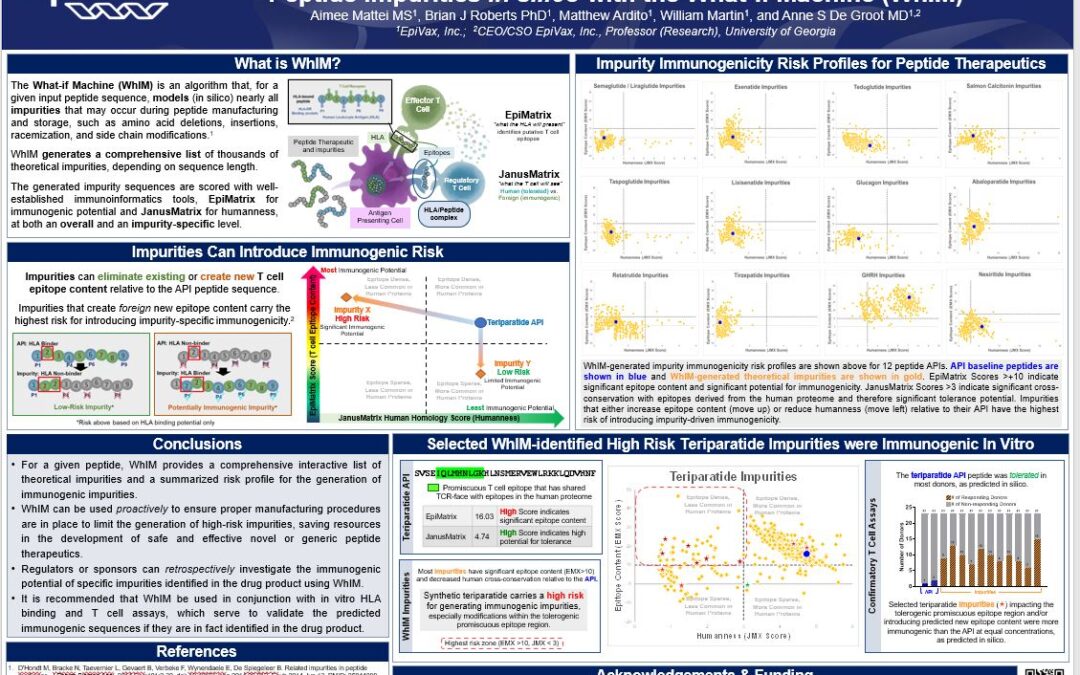
Prospectively Assessing the Immunogenic Risk of Potential Synthetic Peptide Impurities in silico with the What-if Machine (WhIM)
by Elena Iemma | Jul 27, 2023
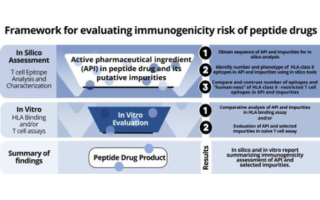
Immunogenicity risk assessment of synthetic peptide drugs and their impurities – ScienceDirect
by Elena Iemma | May 25, 2023
Comparison of Predicted T cell Epitopes in Porcine Circovirus Type 2 Isolates from 2017 to 2021 and Selected Vaccines (EpiCC Analysis) Confirms the Global Relevance of a Bivalent Vaccine Approach