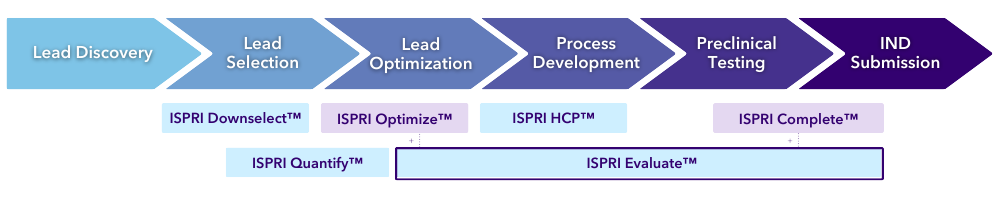
ISPRI Evaluate™
Don’t let immunogenicity surprise you in the clinic.
The ISPRI Evaluate™ report is a comprehensive evaluation of the immunogenic potential of a biologic therapeutic, leveraging EpiVax’s proprietary ISPRI in silico toolkit and key insights from immunogenicity experts. Beginning with a detailed review of your product’s primary amino acid sequence, this analysis delves into the candidate’s overall and regional immunogenic potential through the context of T cell epitope content and regulatory T cell epitope characterization. Regional ‘hotspots’ are identified, and overall risk is compared to similar products with known immunogenicity for benchmarking. The final report will serve multiple purposes throughout the development process, combining objective measures with expert opinions crucial for directing the next phases of R&D.
What can ISPRI Evaluate™ do for you?
-
Provide comprehensive immunogenicity data to include in:
-
Integrated Summary of Immunogenicity (IND-filing preparations)
-
Fundraising materials
-
Publications
-
Research grant applications
-
Internal product characterization/R&D strategy discussions
-
-
Determine the overall and regional immunogenic potential of your lead candidate(s).
-
Rank order lead candidates allowing for intelligent selection and promotion.
-
Direct R&D strategies by reducing volume of downstream in vitro and in vivo immunogenicity studies, saving precious resources (typically 20-fold).
-
Focus optimization on critical protein regions.

Available add-ons:
ISPRI Optimize: EpiVax will make recommendations on which potentially immunogenic regions of the sequence could be good targets for deimmunization.
ISPRI Complete: The team of experts will consider critical aspects of the proposed use and create a detailed report in which all findings are interpreted and put into clinical context, providing a clear roadmap of the next steps in your development process.
Need actionable immunogenicity data?
Learn more about ISPRI Evaluate™!
Looking to screen a large set of candidates for relative immunogenic potential? You need ISPRI Downselect™!
The EpiVax Roadmap