Biotest-EpiVax Collaborative Research Targets New, Non-Immunogenic Treatment for Hemophilia A

Dreieich/ Germany, Rhode Island/ USA, 3 December 2013. Biotest AG, Dreieich, Germany and EpiVax, Inc., Providence, Rhode Island, USA are pleased to announce a new Collaborative Research Agreement. With this collaboration a novel, non-immunogenic Factor VIII (FVIII) should be developed. The coagulation factor VIII used for Hemophilia therapy will be altered in such a way that the immune system of the patients may not respond by developing inhibitory antibodies. The formation of inhibitory antibodies against the Coagulation factor VIII reduces its efficacy and may lead to severe bleeding disorders. Immunogenicity suppression will be also achieved through integration of EpiVax’ proprietary Tregitope, an immune-modulating technology, with the FVIII treatment.
A tolerized and de-immunized version of factor VIII would allow patients and their families to experience the benefits of FVIII treatment, which does not induce a reaction of the immune system (no immunogenicity) without the fear of developing inhibitory antibodies (“inhibitors”) against the therapeutic factor VIII. Studies carried out by EpiVax and collaborators indicate that Tregitope may be useful for reducing the antibody formation (inducing tolerance) to transplants, protein drugs, and allergens by “Tregitope“. This announcement marks the initiation of the joint program, which is in the pre-clinical phase of development.
“This is an entirely novel approach to improving factor VIII therapy. In addition, a whole range of other biologics such as toxins and monoclonal antibodies might also benefit from the same approach.” said Anne De Groot, M.D., President and CEO of EpiVax. “The development of “inhibitors” is a life-threatening complication with a profound impact on patients’ lives. We want to make it possible to avoid the most serious side effect of today’s hemophilia treatment.–” added Jörg Schüttrumpf, M.D., Senior Vice President of Global Research at Biotest.
About Biotest
Biotest is a provider of plasma proteins and biotherapeutic drugs. With a value added chain that extends from pre-clinical and clinical development to worldwide sales, Biotest has specialized primarily in the areas of clinical immunology, hematology and intensive medicine. Biotest develops and markets immunoglobulins, coagulation factors and albumins based on human blood plasma. These are used for diseases of the immune and hematopoietic systems. In addition Biotest develops monoclonal antibodies in the indications of rheumatoid arthritis and cancer of plasma cells, which are produced by recombinant technologies. Biotest has about 1.900 employees worldwide. The preference shares of Biotest AG are listed in the SDAX on the Frankfurt stock exchange.
About EpiVax
EpiVax, Inc., located in Providence, Rhode Island, USA, is a biotechnology company focused on the development of vaccines and biologic therapies. EpiVax is one of the world’s leading innovators in the field of “Immunogenicity Screening”. The company uses proprietary immunoinformatics tools to screen protein therapeutics and to de-immunize these drugs so as to reduce adverse effects in the clinic. The Tregitope technology is an additional immune-modulating tool that is expected to improve tolerance of protein drugs such as replacement enzymes, blood factors, and monoclonal antibodies.
About Tregitopes
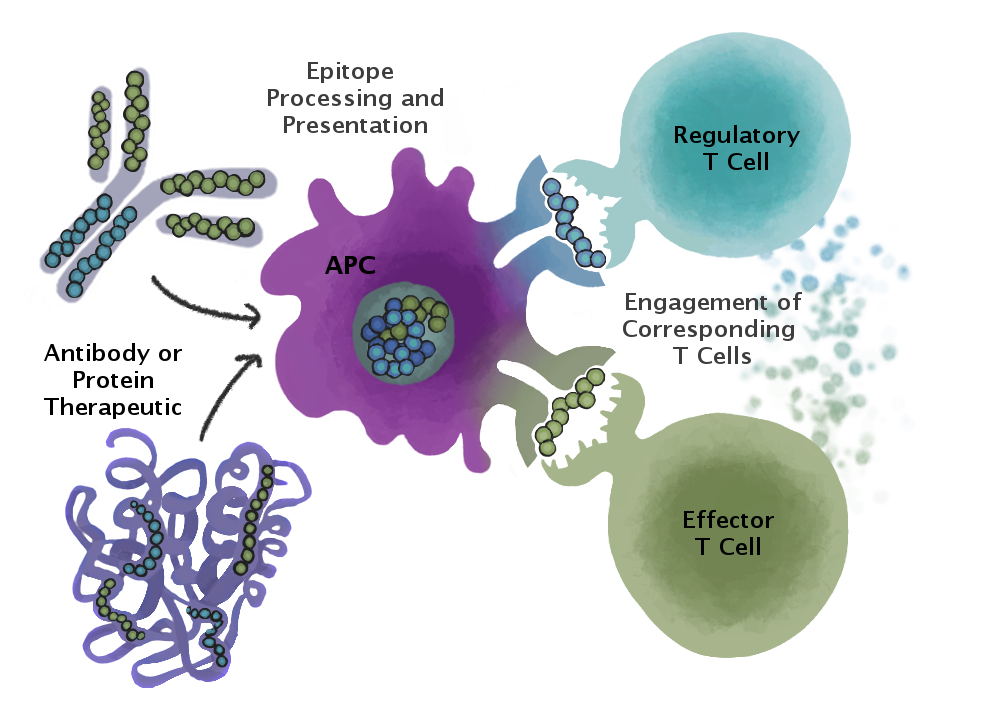 Tregitopes are linear sequences of amino acids contained within the framework of a very common serum protein known as immunoglobulin G. Tregitopes act as a natural immune system ‘off switch” and have been shown in standard preclinical models, to “reset” the immune response away from immunogenicity and towards tolerance. In the current collaboration, EpiVax and Biotest scientists will use Tregitopes to selectively dampening unwanted immune responses to factor VIII, the primary therapeutic used to control bleeding for individuals who have hemophilia A. For more information about Tregitopes visit https://www.epivax.com/pipeline/immune-modulation/.
Tregitopes are linear sequences of amino acids contained within the framework of a very common serum protein known as immunoglobulin G. Tregitopes act as a natural immune system ‘off switch” and have been shown in standard preclinical models, to “reset” the immune response away from immunogenicity and towards tolerance. In the current collaboration, EpiVax and Biotest scientists will use Tregitopes to selectively dampening unwanted immune responses to factor VIII, the primary therapeutic used to control bleeding for individuals who have hemophilia A. For more information about Tregitopes visit https://www.epivax.com/pipeline/immune-modulation/.
About Hemophilia
As a lifelong inherited bleeding disorder, hemophilia affects about 1 in 10,000 people worldwide. Hemophilia is one of a number of such disorders that prevent blood from clotting properly. People with hemophilia experience prolonged internal bleeding that can result from a seemingly minor injury. Bleeding into joints and muscles causes severe pain and disability while bleeding into major organs, such as the brain, can cause death. Treating the bleeding episodes involves the prompt and proper use of clotting factor concentrates. Hemophilia A is caused by a deficiency of clotting factor VIII. Therefore, intravenously administered therapeutic factor VIII is often recognized as a foreign protein (antigen) by the patient´s immune system. As a consequence up to 30% of patients with severe hemophilia develop antibodies against the therapeutic factor VIII. These antibodies are called inhibitors because they reduce or eliminate the therapeutic effect of factor VIII. Most inhibitors develop during early childhood and compromise the ability to effectively prevent or manage hemorrhages, resulting in a greater rate of disability, morbidity, complications and costs of therapy. The formation of inhibitors is the most serious complication of today´s hemophilia treatment. Avoiding the risk of inhibitor development would be the most effective prerequisite for a continuous therapy enabling hemophilia patients to live an almost normal life without irreversible joint damage.
For more information on hemophilia and FVIII, please visit the World Federation of Hemophilia website at https://www.wfh.org/en/page.aspx?pid=646
Acknowledgement
This project builds on research that is currently initially funded by a Small Business Innovation Research (SBIR) grant from the National Heart Lung and Blood Institute (NHLBI), a component of the National Institutes of Health (NIH) (Award Number R43HL114308). These and additional funds from the NIH and private foundations such as JDRF have supported the pre-clinical development of Tregitope applications in hemophilia, diabetes, and biologics. More information about Tregitope applications can be found at this link: Tregitope Applications
Disclaimer
This document contains forward-looking statements on overall economic development as well as on the business, earnings, financial and assets position of Biotest AG and its subsidiaries. These statements are based on current plans, estimates, forecasts and expectations of the company and are thus subject to risks and elements of uncertainty that could result in significant deviation of actual developments from expected developments. The forward-looking statements are only valid at the time of publication. Biotest does not intend to update the forward-looking statements and assumes no obligation to do so.
Biotest AG, Landsteinerstr. 5, 63303 Dreieich, www.biotest.de
Dr. Monika Buttkereit
tel.: +49 (0) 6103 801-4406,
e-mail: investor_relations@biotest.de
fax: +49 (0) 6103 801-347
Securities’ ID No., ISIN ordinary shares: 522720, DE0005227201
Securities’ ID No., ISIN preference shares: 522723, DE0005227235
Listing: Prime Standard
Open Market: Berlin-Bremen, Düsseldorf, Frankfurt, Hamburg, Hannover, München, Stuttgart
EpiVax, Inc. 146 Clifford Street, Providence RI 02903, www.epivax.com
Dr. Annie De Groot MD
e-mail: annied@epivax.com
tel: +1 401-272-2123
mobile: +1 401-952-4227
fax: +1 401-272-7562


