Hello, protein therapeutics developers! We’re presenting some work in Montpellier at the Antibody Industrial Symposium (AIS) and we thought we’d share in advance of the presentation. Why? Because we thought you would want to know that Tregitope content is more important than biophysical properties when developing monoclonal antibodies. In short, Tregitope rocks!
 How did we figure this out? Well, Jain et al (PNAS 2017), put together a very comprehensive database of the biophysical properties of 137 antibodies. More recently, Juan Carlos Almagro published similar data on monoclonal antibodies used for cancer (Frontiers in Immunology, 2017). We analyzed the aggregated data, in which the sequences of 137 antibodies and their biophysical properties was provided, and discovered that Tregitope content, and not humanization nor biophysical properties, was predictive of antibody immunogenicity. For our ISPRI toolkit users, this is great news, because that means that the Tregitope-adjusted scores on the more than 500,000 sequences analyzed by users in the ISPRI toolkit over the past 12 months are extremely predictive of clinical immunogenicity!
How did we figure this out? Well, Jain et al (PNAS 2017), put together a very comprehensive database of the biophysical properties of 137 antibodies. More recently, Juan Carlos Almagro published similar data on monoclonal antibodies used for cancer (Frontiers in Immunology, 2017). We analyzed the aggregated data, in which the sequences of 137 antibodies and their biophysical properties was provided, and discovered that Tregitope content, and not humanization nor biophysical properties, was predictive of antibody immunogenicity. For our ISPRI toolkit users, this is great news, because that means that the Tregitope-adjusted scores on the more than 500,000 sequences analyzed by users in the ISPRI toolkit over the past 12 months are extremely predictive of clinical immunogenicity!
Let me explain: Humanization of monoclonal antibodies is an approach that has been used to render biologics less foreign to the human immune system. Why do biologics developers humanize? Because they are very worried about anti-drug-antibodies (or anti-therapeutic antibodies) which can reduce the efficacy of monoclonals and other protein therapeutics.
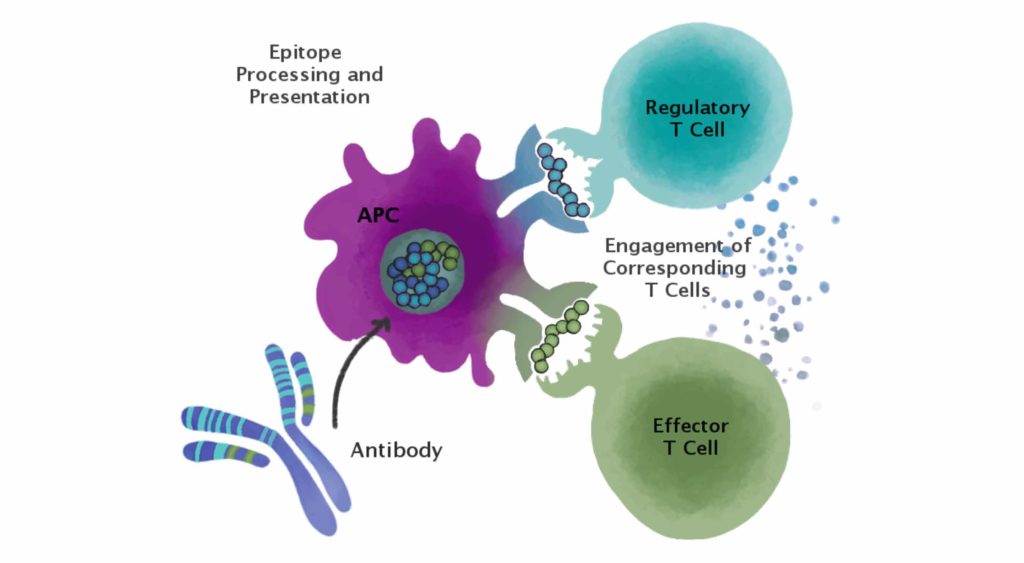
Since (by now) practically everyone knows that T cell epitopes are the key drivers, or modulators, of immunogenicity, EpiVax has developed methods based on T effector and regulatory T cell epitope (Tregitopes) content for evaluating the immunogenic potential of antibodies. We also developed tools for evaluating the potential for epitopes to be conserved with the human genome, now known as JanusMatrix. These tools make it possible to evaluate the potential immunogenicity of monoclonal antibodies, before they are tested in clinical trials.
So what’s new? What’s new is that we could compare “physical properties” of antibodies and immunogenicity risk assessment by our ISPRI toolkit, to see which set of predictors was more likely to be correlated with immunogenicity in the clinic. Given the published information in Jain et al., we were able to analyze three sets of mAbs; (1) 137 mAbs whose key biophysical properties in Jain, and two subsets; (2) 19 mAbs with cancer indications (thank you Juan Carlos Almagro!), and (3) 24 mAbs for which ADA responses were available (these are well characterized and include many of the monoclonals already in the clinic, such as infliximab, secukinumab, etc. The latter two subsets had no overlap.
Each mAb sequence was then screened for the presence of putative effector and regulatory T cell epitopes using the EpiMatrix system on the ISPRI website. To assess the immunogenic potential of each mAb, we calculated (1) Tregitope content, (2) Tregitope-adjusted EpiMatrix score, and (3) the ISPRI-predicted ADA response. To assess the humanness of each mAb, we calculated: (1) the average number of human germline matches per 9-mer peptide, (2) the average frequency of use in a database of human antibodies per 9-mer peptide, (3) the number of matches at >80% amino acid similarity in the human proteome per 9-mer peptide, and (4) the average number of potentially cross-reactive T cell epitope matches (by virtue of conserved T cell receptor (TCR) facing residues and compatible HLA facing residues) per 9-mer peptide. The relationship between the variables of humanness and the variables of predicted immunogenic potential as well as observed ADA response, were assessed using Pearson correlation coefficients. The same calculation was applied to evaluate the relationship between 12 biophysical properties and predicted immunogenic potential and observed ADA response.
What did we find out? Neither biophysical properties nor humanness were correlated with observed ADA response ! This was true set of 24 mAbs with known immunogenicity, and in addition, in the other datasets, we did not find any significant correlation between biophysical properties or humanness and our metrics of immunogenic potential. We found that effector T cell epitope content adjusted by Tregitope content (the Tregitope-Adjusted EpiMatrix Score) and the EpiMatrix predicted ADA response were significantly correlated with observed ADA response. In conclusion, Tregitope-adjusted EpiMatrix Score is a better predictor of antibody immunogenicity than either humanness or biophysical properties.
Look for us in Montpelier in June and we’ll be happy to discuss over aperitifs!